Silicate Mineral Structure
The Silicate minerals group is of great importance because they constitute about 90% of the Earth’s crust. They are found in all the common rocks except limestone. To understand the differences between major silicate mineral groups, it is necessary to study their structure. Every silicate mineral contains oxygen and silicon, and all except quartz, contain one or more additional elements to complete their structure.
The basic unit in all silicate minerals is the “silicon-oxygen tetrahedron”. This structure is composed of four oxygen atoms with the silicon atom at its centre. These tetrahedra can occur in the silicate structures either as single units or joined into chains, sheets, and three-dimensional networks by sharing oxygen atoms. Depending upon the type of structure built by these tetrahedra, the slicates are classified into the following groups:
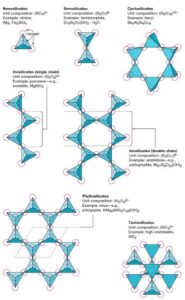
- Nesosilicates. Nesosilicates include those minerals which are built up from isolated SiO4 tetrahedra. The atomic packing of the neosilicate structure is generally dense, which causes the minerals of this to have relatively high specific gravity and hardness. The crystal habit of these minerals is generally equidimensional, and they have poor cleavage. Olivine, Zircon, and Gernets are examples of this class.
- Sorosilicate. Sorosilicates are characterised by a linked pair of SiO4 tetrahedra. In this structure, only one oxygen is shared, giving the ratio of Si : O =2:7. Hemimorphite [Zn4 (Si2O7) (OH)2 H2O] is an example of this class.
- Cyclosilicate. Cyclosilicate is also known as the ring silicate. It contains rings of linked SiO4 tetrahedra having a ratio of Si:O = 1:3. These rings may consist of groups of three, four or six linked tetrahedra. Cyclosilicates from extremely strong minerals, such as beryl and tourmaline.
- Inosilicate (Chain Silicate). In this group, SiO4 tetrahedra are linked by sharing oxygens to form straight chains of indefinite length. These chains may be single chains or a double-linked channel. In the single chain structure, two of the four oxygens in each SiO4 tetrahedron are shared, giving a ratio of Si:O = 1:3. In the double chain structure, half of the tetrahedra share three oxygens, while the other half share two oxygens yielding a ratio of Si:0 =4:11. Inosilicates split easily in one crystal direction because bonds within chains are strong but are weaker between them. These minerals commonly form needle-like crystals, such as asbestos. Pyroxenes are examples of single-chain minerals, and amphiboles are examples of double-chain minerals.
- Phylosilicates (Sheet Silicates). The phylosilicates form sheet structures in which there is the continuous linking of hexagonal groups of silica tetrahedra. In this structure, three of the four oxygens in each SiO4 tetrahedron are shared with neighbouring tetrahedra, giving a ratio of Si:O =2:5. As the atomic bonding perpendicular to the sheet structure is generally weak, these minerals split easily into thin sheets. Flaky minerals, such as micas, chlorite and kaolinite, are examples of this class.
- Tectosilicates. They are also known as the framework silicates. In tectosilicates, SiO4 tetrahedra are linked in a three-dimensional framework. All oxygens in each SiO4 tetrahedron are shared with neighbouring tetrahedra. This results in a strongly bonded structure in which the ratio Si:O is 1:2. The minerals belonging to the tectosilicate group possess uniform properties throughout. Quartz are felspars are the examples of this class.
Table showing Silicate Mineral Groups and Examples.
| Major group | Structure | Chemical formula | Example |
|---|---|---|---|
| Nesosilicates | isolated silicon tetrahedra | [SiO4]4− | olivine, garnet, zircon… |
| Sorosilicates | double tetrahedra | [Si2O7]6− | epidote, melilite group |
| Cyclosilicates | rings | [SinO3n]2n− | beryl group, tourmaline group |
| Inosilicates | single chain | [SinO3n]2n− | pyroxene group |
| Inosilicates | double chain | [Si4nO11n]6n− | amphibole group |
| Phyllosilicates | sheets | [Si2nO5n]2n− | micas and clays |
| Tectosilicates | 3D framework | [AlxSiyO(2x+2y)]x− | quartz, feldspars, zeolites |
List of Silicate Minerals
01. Nesosilicates or Orthosilicates
Nesosilicates (word derived from ancient Greek language named after “Nesos Island”), also known as Orthosilicates, have the orthosilicate ion, present as isolated (insular) [SiO4]4− tetrahedra connected only by interstitial cations. The Nickel–Strunz classification is 09.A. Following minerals and mineral groups are included in this category. Minerals of this group along with their chemical structure are given as under;
- Phenakite group
- Phenakite – Be2SiO4
- Willemite – Zn2SiO4
- Olivine group
- Forsterite – Mg2SiO4
- Fayalite – Fe2SiO4
- Tephroite – Mn2SiO4
- Garnet group
- Pyrope – Mg3Al2(SiO4)3
- Almandine – Fe3Al2(SiO4)3
- Spessartine – Mn3Al2(SiO4)3
- Grossular – Ca3Al2(SiO4)3
- Andradite – Ca3Fe2(SiO4)3
- Uvarovite – Ca3Cr2(SiO4)3
- Hydrogrossular – Ca3Al2Si2O8(SiO4)3−m(OH)4m
- Zircon group
- Zircon – ZrSiO4
- Thorite – (Th,U)SiO4
- Hafnon – (Hf,Zr)SiO4
- Al2SiO5 group
- Andalusite – Al2SiO5
- Kyanite – Al2SiO5
- Sillimanite – Al2SiO5
- Dumortierite – Al6.5–7BO3(SiO4)3(O,OH)3
- Topaz – Al2SiO4(F,OH)2
- Staurolite – Fe2Al9(SiO4)4(O,OH)2
- Humite group – (Mg,Fe)7(SiO4)3(F,OH)2
- Norbergite – Mg3(SiO4)(F,OH)2
- Chondrodite – Mg5(SiO4)2(F,OH)2
- Humite – Mg7(SiO4)3(F,OH)2
- Clinohumite – Mg9(SiO4)4(F,OH)2
- Datolite – CaBSiO4(OH)
- Titanite – CaTiSiO5
- Chloritoid – (Fe,Mg,Mn)2Al4Si2O10(OH)4
- Mullite (aka Porcelainite) – Al6Si2O13
02. Sorosilicates
Sorosilicates (from the Greek word Sōros ‘heap, mound’) have isolated pyrosilicate anions Si2O6−7. It consists of double tetrahedra with a shared oxygen vertex—a silicon: oxygen ratio of 2:7. The Nickel–Strunz classification is 09.B.
Examples include:
- Thortveitite (Sc,Y)2(Si2O7)
- Hemimorphite (calamine) Zn4(Si2O7)(OH)2·H2O
- Lawsonite CaAl2(Si2O7)(OH)2·H2O
- Axinite (Ca,Fe,Mn)3Al2(BO3)(Si4O12)(OH)
- Ilvaite CaFeII2FeIIIO(Si2O7)(OH)
- Epidote group (has both (SiO4)4− and (Si2O7)6− groups}
- Epidote Ca2(Al,Fe)3O(SiO4)(Si2O7)(OH)
- Zoisite Ca2Al3O(SiO4)(Si2O7)(OH)
- Tanzanite – Ca2Al3O(SiO4)(Si2O7)(OH)
- Clinozoisite – Ca2Al3O(SiO4)(Si2O7)(OH)
- Allanite – Ca(Ce,La,Y,Ca)Al2(FeII,FeIII)O(SiO4)(Si2O7)(OH)
- Dollaseite-(Ce) – CaCeMg2AlSi3O11F(OH)
- Vesuvianite (idocrase) – Ca10(Mg,Fe)2Al4(SiO4)5(Si2O7)2(OH)4
03. Cyclosilicates
Cyclosilicates (from Greek word “Kýklos” ‘circle’), or ring silicates, have three or more tetrahedra linked in a ring. The general formula is (SixO3x)2x−, where one or more silicon atoms can be replaced by other 4-coordinated atom(s). The silicon: oxygen ratio is 1:3. Double rings have the formula (Si2xO5x)2x− or a 2:5 ratio. The Nickel–Strunz classification is 09.C. Possible ring sizes include:
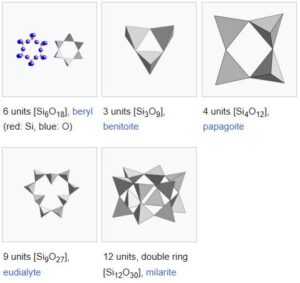
Some example minerals are:
- 3-member single ring
- Benitoite – BaTi(Si3O9)
- 4-member single ring
- Papagoite – CaCuAlSi2O6(OH)3.
- 6-member single ring
- Beryl – Be3Al2(Si6O18)
- Bazzite – Be3Sc2(Si6O18)
- Sugilite – KNa2(Fe,Mn,Al)2Li3Si12O30
- Tourmaline – (Na,Ca)(Al,Li,Mg)3–(Al,Fe,Mn)6(Si6O18)(BO3)3(OH)4
- Pezzottaite – Cs(Be2Li)Al2Si6O18
- Osumilite – (K,Na)(Fe,Mg)2(Al,Fe)3(Si,Al)12O30
- Cordierite – (Mg,Fe)2Al4Si5O18
- Sekaninaite – (Fe+2,Mg)2Al4Si5O18
- 9-member single ring
- Eudialyte – Na15Ca6(Fe,Mn)3Zr3SiO(O,OH,H2O)3(Si3O9)2(Si9O27)2(OH,Cl)2
- 6-member double ring
- Milarite – K2Ca4Al2Be4(Si24O60)H2O
The ring in axinite contains two B and four Si tetrahedra and is highly distorted compared to the other 6-member ring cyclosilicates.
04. Single Chaine Inosilicates
Single-chain inosilicates are a group of silicate minerals where silica tetrahedra (SiO4)(\text{SiO}_4) are linked together in continuous single chains. These are most commonly known as the pyroxene group.
- Pyroxene group
- Clinopyroxene subgroup
- Aegirine (or acmite) – NaFe3+Si2O6
- Augite – (Ca,Mg,Fe)2Si2O6
- Diopside – CaMgSi2O6
- Hedenbergite – CaFe2+Si2O6
- Jadeite – Na(Al,Fe3+)Si2O6
- Pigeonite – (CaxMgyFez)(Mgy1Fez1)Si2O6, where 0.1 ≤ x ≤ 0.4, x + y + z = 1 and y1 + z1 = 1
- Spodumene – LiAlSi2O6
- Orthopyroxene subgroup
- Enstatite – Mg2Si2O6
- Ferrosilite – Fe2+2Si2O6
- Clinopyroxene subgroup
- Pyroxferroite – (Fe,Mn,Ca)SiO3
- Rhodonite – CaMn3Mn(Si5O15)
- Wollastonite group
- Pectolite – NaCa2Si3O8(OH)
- Wollastonite – Ca3(Si3O9)
05. Double Chain Inosilicates
Double-chain inosilicates are silicate minerals where silica tetrahedra (SiO4)(\text{SiO}_4) link together to form paired (double) chains. These are characteristic of the amphibole group.
- Amphibole group
- Anthophyllite – (Mg,Fe)7Si8O22(OH)2
- Cummingtonite series
- Cummingtonite – Fe2Mg5Si8O22(OH)2
- Grunerite – Fe7Si8O22(OH)2
- Tremolite series
- Tremolite – Ca2Mg5Si8O22(OH)2
- Actinolite – Ca2(Mg,Fe)5Si8O22(OH)2
- Hornblende – (Ca,Na)2–3(Mg,Fe,Al)5Si6(Al,Si)2O22(OH)2
- Sodium amphibole group
- Glaucophane – Na2Mg3Al2Si8O22(OH)2
- Riebeckite (asbestos) – Na2FeII3FeIII2Si8O22(OH)2
- Arfvedsonite – Na3(Fe,Mg)4FeSi8O22(OH)2

06. Phyllosilicates
Phyllosilicates (also called sheet silicates) are minerals in which silica tetrahedra (SiO4)(\text{SiO}_4) are linked to form two-dimensional sheets. The name comes from the Greek “Phyllon” meaning leaf, reflecting their layered structure. Basic structure;
Each tetrahedron shares three oxygen atoms with neighbouring tetrahedra, creating wide, flat sheets.
(Si2O5)n(Si_2O_5)_n
- The repeating unit is (Si₂O₅)ₙ
- The fourth oxygen points out of the sheet, allowing bonding with other layers (like octahedral sheets of Al, Mg, or Fe)
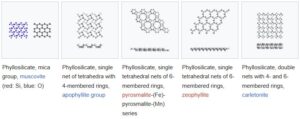
Minerals included in this group are:
- Ajoite (K,Na)Cu7AlSi9O24(OH)6·3H2O
- Apophyllite group
- Fluorapophyllite-(K) KCa4(Si8O22)F·8H2O
- Bannisterite (Ca,K,Na)(Mn2+,Fe2+)10(Si,Al)16O38(OH)8·nH2O
- Carletonite KNa4Ca4Si8O18(CO3)4(OH,F)·H2O
- Cavansite Ca(VO)Si4O10·4H2O (dimorph of pentagonite)
- Chlorite group – (Al,Fe2+,Fe3+Li,Mg,Mn,Ni)5−6(Al,Fe3+,Si)4(O,OH)18 (2:1:1 clays)
- Chamosite – (Fe2+,Mg,Al,Fe3+)6(Si,Al)4O10(OH,O)8 (Fe endmember)
- Clinochlore – Mg5Al(AlSi3O10)(OH)8 (Mg endmember)
- Cookeite – (LiAl4◻)[AlSi3O10](OH)8
- Chrysocolla Cu2−xAlx(H2−xSi2O5)(OH)4·nH2O, x < 1
- Ekanite Ca2ThSi8O20
- Gyrolite NaCa16Si23AlO60(OH)8·14H2O
- Hisingerite Fe3+2(Si2O5)(OH)4·2H2O
- Imogolite Al2SiO3(OH)4
- Kaolinite-Serpentine group
- Greenalite (Fe2+,Fe3+)2−3Si2O5(OH)4
- Kaolinite subgroup (1:1 clays)
- Dickite – Al2(Si2O5)(OH)4
- Kaolinite – Al2Si2O5(OH)4
- Halloysite – Al2Si2O5(OH)4
- Serpentine subgroup
- Amesite Mg2Al(AlSiO5)(OH)4
- Antigorite Mg3Si2O5(OH)4
- Chrysotile Mg3Si2O5(OH)4
- Lizardite Mg3Si2O5(OH)4
- Mica group
- Brittle mica group
- Clintonite – CaAlMg2(SiAl3O10)(OH)2
- Margarite – CaAl2(Al2Si2)O10(OH)2
- Dioctahedral mica group
- Celadonite subgroup
- Celadonite – K(MgFe3+◻)(Si4O10)(OH)2
- Glauconite K0.60−0.85(Fe3+,Mg,Al)2(Si,Al)4O10](OH)2
- Muscovite KAl2(AlSi3)O10(OH)2
- Paragonite NaAl2(AlSi3O10)(OH)2
- Roscoelite – K(V3+,Al)2(AlSi3O10)(OH)2
- Celadonite subgroup
- Trioctahedral mica group
- Aspidolite – NaMg3(AlSi3O10)(OH)2
- Biotite subgroup K(Fe2+,Mg)2(Al,Fe3+,Mg,Ti)([Si,Al,Fe]2Si2O10)(OH,F)2
- Annite KFe2+3(AlSi3O10)(OH)2 (Fe endmember)
- Phlogopite KMg3(AlSi3)O10(OH)2 (Mg endmember)
- Lepidolite (polylithionite-trilithionite series) – K(Li2,Li1.5Al1.5)AlSi3−4O10(F,OH)2
- Zinnwaldite series – KFe2+2Al(Al2Si2O10)(OH)2
- Brittle mica group
- Neptunite KNa2Li(Fe2+)2Ti2[Si4O12]2
- Okenite Ca10Si18O46·18H2O
- Palygorskite group (2:1 clays)
- Palygorskite Al2Mg2◻2Si8O20(OH)2(H2O)4·4H2O
- Tuperssuatsiaite Fe3+Fe3+2(Na◻)◻2Si8O20(OH)2(H2O)4·2H2O
- Pentagonite Ca(VO)Si4O10·4H2O (dimorph of cavansite)
- Pyrophyllite-Talc group
- Pyrophyllite Al2Si4O10(OH)2
- Talc Mg3Si4O10(OH)2 (2:1 clay)
- Sepiolite group
- Sepiolite Mg4(Si6O15)(OH)2·6H2O (2:1 clay)
- Falcondoite (Ni,Mg)4Si6O15(OH)2·6H2O (Ni analogue of sepiolite)
- Smectite group (2:1 clays)
- Hectorite Na0.3(Mg,Li)3(Si4O10)(F,OH)2
- Montmorillonite (Na,Ca)0.33(Al,Mg)2(Si4O10)(OH)2·nH2O
- Nontronite Na0.3Fe2((Si,Al)4O10)(OH)2·nH2O
- Saponite Ca0.25(Mg,Fe)3((Si,Al)4O10)(OH)2·nH2O
- Stevensite (Ca,Na)xMg3−x(Si4O10)(OH)2
- Stilpnomelane group
- Stilpnomelane (K,Ca,Na)(Fe,Mg,Al)8(Si,Al)12(O,OH)36·nH2O
- Vermiculite Mg0.7(Mg,Fe,Al)6(Si,Al)8O20(OH)4·8H2O (2:1 clay)
07. Tectosilicates
Tectosilicates (also called framework silicates) are the most structurally complex silicate minerals. In this group, every silica tetrahedron (SiO4)(\text{SiO}_4) shares all four of its oxygen atoms with neighboring tetrahedra, forming a 3D interconnected framework.
Minerals included in this group are;
- Quartz group (silica) SiO2
- Chalcedony – a cryptocrystalline variety of silica composed mostly of quartz with some moganite
- Polymorphs of silica
- α-quartz – trigonal, “normal” quartz under 573 °C (846 K; 1,063 °F)
- β-quartz – hexagonal, high-temperature quartz
- Coesite – monoclinic
- Cristobalite – tetragonal
- Melanophlogite – cubic or tetragonal, rare
- Moganite – monoclinic
- Stishovite – tetragonal, extremely hard and dense
- Tridymite – orthorhombic
- Feldspar group
- Alkali feldspar series (potassium feldspars or K-spar)
- Microcline KAlSi3O8
- Orthoclase KAlSi3O8
- Anorthoclase (Na,K)AlSi3O8
- Sanidine KAlSi3O8
- Plagioclase feldspar series
- Albite NaAlSi3O8 (Na endmember)
- Oligoclase (Na,Ca)Al(Si,Al)Si2O8 (Na:Ca 90:10 to 70:30)
- Andesine (Na,Ca)Al(Si,Al)Si2O8 (Na:Ca 50:50 to 70:30)
- Labradorite (Ca,Na)Al(Al,Si)Si2O8 (Na:Ca 30:70 to 50:50)
- Bytownite (Ca,Na)Al(Al,Si)Si2O8 (Na:Ca 10:90 to 30:70)
- Anorthite CaAl2Si2O8 (Ca endmember)
- Other feldspars
- Buddingtonite NH4AlSi3O8
- Celsian BaAl2Si2O8
- Hyalophane (K,Ba)[Al(Si,Al)Si2O8]
- Alkali feldspar series (potassium feldspars or K-spar)
- Feldspathoid group
- Cancrinite subgroup
- Afghanite (Na,K)22Ca10[Si24Al24O96](SO4)6Cl6
- Cancrinite (Na,Ca,◻)8(Al6Si6O24)(CO3,SO4)2·2H2O
- Sacrofanite (Na61K19Ca32)(Si84Al84O336)(SO4)26Cl2F6·2H2O
- Leucite K(AlSi2O6)
- Nepheline subgroup
- Nepheline – Na3K(Al4Si4O16)
- Sodalite subgroup
- Hauyne Na3Ca(Si3Al3)O12(SO4)
- Lazurite Na7Ca(Al6Si6O24)(SO4)(S3)·H2O
- Nosean Na8(Al6Si6O24)(SO4)·H2O
- Sodalite Na4(Si3Al3)O12Cl
- Tugtupite Na4(BeAlSi4O12)Cl
- Cancrinite subgroup
- Scapolite group
- Marialite Na4Al3Si9O24Cl
- Meionite Ca4Al6Si6O24CO3
- Zeolite group
- Amicite K2Na2Al4Si4O16·5H2O
- Analcime Na(AlSi2O6)·H2O
- Brewsterite subgroup (Ba,Sr,Ca)Al2Si6O16·5H2O
- Chabazite-Lévyne subgroup
- Chabazite – M[Al2Si4O12]·6H2O
- Lévyne (Ca1−2,Na1−2,K2)Al2Si4O12·6H2O
- Clinoptilolite subgroup (Na,Ca,K)3−6(Al6−7Si29−30O72)·20H2O
- Erionite subgroup (Na1−2,K1−2,Ca1−2)2Al4Si14O36·15H2O
- Faujasite subgroup (Na1−2,Ca1−2,Mg1−2)3.5[Al7Si17O48]·32H2O
- Ferrierite subgroup [Mg2(K,Na)2Ca0.5](Si29Al7)O72·18H2O (Ferrierite-Mg)
- Heulandite subgroup (Na,Ca,K)5−6[Al8−9Si27−28O72]·nH2O
- Laumontite CaAl2Si4O12·4H2O
- Mordenite (Na2,Ca,K2)4(Al8Si40)O96·28H2O
- Natrolite subgroup
- Mesolite – Na2Ca2Si9Al6O30·8H2O
- Natrolite – Na2Al2Si3O10·2H2O
- Scolecite – CaAl2Si3O10·3H2O
- Paulingite subgroup (K2,Ca,Na2,Ba)5[Al10Si35O90]·45H2O (Paulingite-K)
- Phillipsite subgroup
- Phillipsite – (Ca3(Si10Al6)O32·12H2O (Phillipsite-Ca)
- Pollucite (Cs,Na)2(Al2Si4O12)·2H2O
- Stilbite subgroup
- Stellerite – Ca4(Si28Al8)O72·28H2O
- Stilbite – (NaCa4,Na9)(Si27Al9)O72·28H2O
- Thomsonite subgroup – NaCa2Al5Si5O20·6H2O (Thomsonite-Ca)
- Yugawaralite CaAl2Si6O16·4H2O