Physical Properties of Mineral
One can readily determine the physical properties of the minerals merely by inspection or simple tests. Because the physical properties are determined in hand specimens, they are important in the recognition of minerals in the field. The chief physical properties are colour, streak, lustre, hardness, habit, cleavage, fracture, odour, feel, tenacity, fluorescence, phosphorescence, magnetism, specific gravity, and crystal forms.
The correct identification of minerals is made with the help of a polarising microscope. This involves grinding the minerals or rocks into very thin slices and allowing polarised light to pass through them. In this way, their optical properties are studied, and the minerals are identified. Opaque minerals such as ores are studied under the ore-microscope in the reflected light.
01. Colour of Minerals
The colour of a mineral is due to the absorption of certain wavelengths of light by atoms making up the crystal. The remaining wavelengths of white light that are not absorbed give rise to the colour seen by the observer. Thus, dark coloured minerals absorb most of the light, whereas red minerals reflect or transmit mainly the red light and absorb all others.
Some minerals possess characteristic and fairly constant colour, for example, the lead-grey of galena, bass-yellow of pyrite, and green of chlorite. But in other cases, such as quartz, the colour is variable and can not be relied on as a guide to identify minerals.
Presence of a small amount of impurities can give a variety of colours to a white or colourless mineral. For example, the colour of amethyst and rose quartz is due to the presence of titanium or manganese in traces. The most common colouring impurity is hematite. It imparts red colour to many minerals, including some felspar, calcite, and jasper.
Some minerals, when viewed in different directions, show irregular changes in colour tints. This is called “play of colour”. The term “opalescence” is applied to minerals which show milky appearance, for example, opal. When bands of prismatic colours are seen on the surface of a mineral, it is said to show “iridescence”.
02. Streak of Minerals
The colour of the mineral powder is called streak. It is more consistent and reliable than the body colour of the mineral. The streak is obtained by rubbing a mineral against an unglazed porcelain plate, called the “streak plate”. The study of streak is most useful in the case of coloured minerals, which often give a much lighter streak than their body colour. For example, hematite, which appears almost black, gives a red coloured streak. However, the streak is less useful for identifying most of the silicates, carbonates and transparent minerals because they give a white streak.
03. Lustre of Minerals
Lustre is a very characteristic and useful property of minerals. It is a measure of the reflectivity of the mineral surface. The lustre may be defined as the general appearance of a mineral surface in reflected light. The various types of lustre are as follows.
- Metallic Lustre. Minerals which have the appearance of a metal are said to have a “metallic lustre”. e.g. pyrite, and galena.
- Submatallic Lustre. The feebly displayed metallic lustre is called the “submattalic lustre”, eg. chromite and hematite.
- Adamantine Lustre. A hard, brilliant lustre like that of a diamond is called “adamantine lustre”. It is due to the mineral’s high index of refraction, e.g. transparent cerssite.
- Vitreous Lustre. It is the lustre exhibited by the broken glass, e.g. quartz.
- Pearly Lustre. It is the lustre exhibited by the pearls, e.g. muscovite, talk, and calcite.
- Silky Lustre. It is the lustre exhibited by the silk fibres. Minerals which crystallize the fibrous habit commonly show silky lustre, e.g., asbestos and fibrous gypsum.
- Resinous Lustre. It is the lustre exhibited by the resin, e.g. sphalerite.
- Greasy Lustre. It is the lustre exhibited by the grease, e.g. talc, and nephaline.
- Dull or Earth Lustre. Minerals showing no lustre are said to possess dull or earthy lustre. e.g. kaolin.
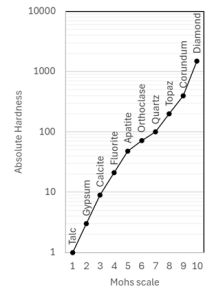
04. Hardness of Minerals
Hardness is one of the most useful diagnostic properties of a mineral. It is defined as the resistance of a mineral to abrasion or scratching. Hardness is determined by rubbing a mineral of unknown hardness against one of known hardness. A numerical value is obtained by using the “Mohs Scale of hardness”. On the scale, there are ten minerals which are arranged in order of their increasing hardness.
In the absence of hardness testing minerals, the following materials may be used to determine approximate hardness. (i) A finger nail will scratch up to about 2.5 (i.e. not calcite), (ii) A window glass will scratch up to about 5 (i.e not felspar), (iii) A penknife will scratch up to about 6.5 (i.e. not quarts).
05. Cleavage of Minerals
If a mineral breaks along a flat plane, it is said to possess a cleavage; if it breaks with an irregular surface, it is said to show a fracture. Thus, the cleavage may be defined as the tendency of a mineral to break more easily with smooth surfaces along plates of weak bonding. Hence, cleavage is the property which is related to the atomic arrangement within the mineral. Because the cleavage always occurs parallel to the possible crystal face, it is octahedral, prismatic, abasal and so on. For example, galena has cubic cleavage, fluorite has octahedral cleavage, mica has basal cleavage, and calcite has rhombohedral cleavage.
Depending on the ease with which a crystal cleavages and the perfection of the surface obtained, the cleavage is classified as “perfect”, “good”, “poor”, and “indistinct”. Examples of minerals which show perfect cleavage are mica, galena, calcite, and fluorite. Quartz has no cleavage at all.
06. Fracture of Minerals
Minerals which do not exhibit cleavage break with an irregular surface. The nature of this broken surface is called “fracture”. In case of fracture, the breaking should be in any other direction than the cleavage. Unlike cleavage, the fracture does not produce a smooth surface. The most common types of fracture are as follows;
- Conchoidal Fracture. It is a curved fracture surface showing concentric lines like a shell. Quartz and glass show conchoidal fracture.
- Even Fractrue. It is a fracture surface which is almost flat. Flint shows even fractures.
- Uneven Fracture. It is a fractured surface which is irregular and rough. A large number of minerals show uneven fracture.
- Hacky fracture. It is a fracture surface which is rough with sharp and jagged points. Native metals show backly fracture.
07. Odour of Minerals
Some minerals give a characteristic smell when rubbed, breathed upon or heated. The chief types of odour are as follows:
- Arsenical. The arsenical odour is like the odour of garlic. Orpiment and other arsenic minerals give arsenical odour.
- Sulfurous. This odour is like the odour of burning sulfur. Pyrite gives a sulfurous odour.
- Argillaceous. This odour is like the odour of clay. Kaloin gives an argillaceous odour.
08. Feel of Minerals
Feel is the sensation upon touching or handling minerals. The different types of feel are greasy, soapy, rough, and harsh. The example of a greasy feel is talc, that of a soapy feel is Kaoline, and that of a rough feel is bauxite.
09. Tenacity of Minerals
Tenacity of a mineral denotes the degree or character of cohesion. Tenacity is classified as follows.
- Sectile. Minerals which may be cut with a knife, but slices are not malleable.
- Malleable. Minerals which flatten under the hammer.
- Flexible. Minerals which may be bent.
- Elastic. Minerals which spring back after bending.
- Brittle. Minerals which break easily on bending. Brittle is the opposite of tough.
- Friable. Minerals which crumble easily.
- Pulverulent. Minerals which are powdery and have little or no cohesion, e.g. clay or chalk.
10. Fluorescence of Minerals
Some minerals, when exposed to sunlight or ultraviolet light, produce a colour quite different from their own. Thus, green or colourless fluorite shows a blue or purple colour in ultraviolet light. This property of minerals is called “fluorescence”. The other minerals which often show fluorescence are calcite and scheelite.
11. Phosphorescence
Some minerals glow and emit light when they are placed in ultraviolet light or certain other electrical radiation. The glow induced in the mineral may continue for a few seconds or minutes after the removal of the cause. This property of minerals is called phosphorescence. The examples of minerals which show phosphorescence are diamond and sphalerite.
12. Magnetism
A few minerals are attracted by magnets. Of these minerals, magnetite and pyrite are the most common examples. The magnetite that possesses attracting power and polarity is called “lodestone”.
13. Habit of a Mineral
The habit of a mineral may be defined as the size and shape of the crystals, and the structure or form shown by the crystal aggregates and cryptocrystalline masses. The chief habits shown by minerals are as follows.
- Accicular. Minerals showing needle-like crystals, for example, natrolite.
- Fibrous. Minerals showing an aggregate of long thin fibres, for example, asbestose, and satinspar.
- Foliated. Minerals with a platy habit commonly occur as foliated aggregates, containing thin, separable sheets, for example, muscovite and biotite.
- Bladed. Minerals showing a bladed habit occur as small knife blades, for example, kyanite.
- Tabular. Minerals showing broad flat surfaces, for example, felspar.
- Columnar. Minerals showing columnar crystals, for example, tourmaline. The term stalactitic refers to columnar forms of minerals, such as calcite and aragonite.
- Botryoidal. Minerals showing aggregate of rounded masses resembling a bunch of grapes, e.g. chalcedony.
- Reniform. Minerals showing kidney-shaped form, e.g. kidney iron ore.
- Granular. Minerals which occur as aggregates of equidimensional grains, for example, chromite.
- Pisolitic. Minerals which occur as aggregates of rounded grains of a pea-sized, for example, bauxite.
- Oolitic. Minerals showing an aggregate of bodies resembling fish roe. In this case, the rounded grains are the size of a small pinhead.
- Massive. When noncrystalline or cryptocrystalline minerals occur as a structureless mass, their habit is described as massive, for example, flint.
See: Chemical Properties of Minerals









